Search
How Much Are DePuy ASR Cases Really Worth?
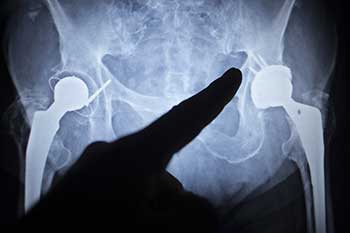 All civil lawsuits come down to one thing— money. Sure, there will always be people who claim that they have filed their lawsuit out of a a sense of social responsibility and a genuine desire to help mankind, but injury cases– and medical device cases in particular– are really about getting financial compensation for an injury.
All civil lawsuits come down to one thing— money. Sure, there will always be people who claim that they have filed their lawsuit out of a a sense of social responsibility and a genuine desire to help mankind, but injury cases– and medical device cases in particular– are really about getting financial compensation for an injury.
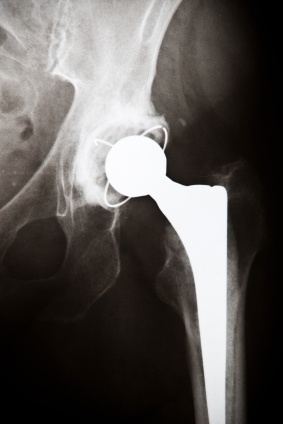
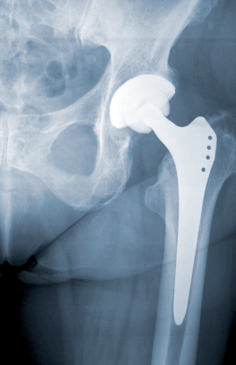
As soon as the DePuy ASR hip prosthesis was pulled from the market in August, 2010, a stream of people with these defective metal hip implants filed lawsuits against the manufacturer alleging amongst several things that the device resulted in pain, disability which may had resulted in the need for additional medical care or perhaps prevented a person from going about their employment.
 Nursing Home Law News
Nursing Home Law News